element group 1
element group 1

Group 1 - physical properties - Groups in the periodic
Group 1 contains elements placed in a vertical column on the far left of the periodic table. The elements in group 1 are called the alkali metals. Group 1 is on the left-hand side of the
Learn More
The Parts of the Periodic Table - Angelo State University
Group 1A (or IA) of the periodic table are the alkali metals: hydrogen (H), lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr).These are (except for hydrogen) soft, shiny, low-melting, highly reactive metals, which tarnish when exposed to air. The name comes from the fact that when these metals or their oxides are dissolved in water, a basic (alkaline
Learn More
Reactions of Group 1 Elements with Water - Chemistry LibreTexts
Aug 15, · All of Group 1 elements —lithium, sodium, potassium, rubidium and cesium react vigorously or even explosively with cold water. In each case, the aqueous metal hydroxide and hydrogen gas are produced, as shown: (1) 2 X ( s) + 2 H 2 O ( l) → 2 X O H ( a q) + H 2 ( g) where X is any Group 1 metal.
Learn More
Materials Testing Laboratory Locations | Element
One of the largest and fastest growing companies in the testing sector, Element's laboratories span the US, UK, Netherlands, Belgium, Germany, Spain & Asia.
Learn More
Periodic Table of Elements - PubChem
Finally, IUPAC assigns collective names (lanthanoids and actinoids) and group numbering (1 to 18) and has investigated the membership of the group 3 elements. PubChem is working with IUPAC to help make information about the elements and the periodic table machine-readable.
Learn More
Group 1 (1A) - Alkali Metals - umb.edu
element unique chemistry. Because beryllium's chemistry is so different from the other group 2 elements, the term "alkaline earth" is usually restricted to Mg, Ca, Sr, Ba, Ra.!Mg2+ has a similar charge density to Li+, so the two elements show some similar chemistry (e.g., tendency to form useful organometallic compounds).
Learn More
The chemical elements of the periodic table sorted by density - Lenntech
Chemical elements listed by density The elements of the periodic table sorted by density. click on any elements name for further chemical properties, Phone: +1 877 453 8095 e-mail: [email protected]. Lenntech DMCC (Middle East) Level 6 - OFFICE #101-One JLT Tower Jumeirah Lake Towers Dubai - U.A.E.
Learn More
Hydrogen and Alkali Metals | CK-12 Foundation
Group 1 of the periodic table includes hydrogen and the alkali metals. · Because they have just one valence electron, group 1 elements are very
Learn More
What are the Group 1 elements called? - Q & A - BYJUS
The six elements of Group 1 in the periodic table are: Lithium (Li) Sodium (Na) Potassium (K) Rubidium (Rb) Cesium (Cs) Francium (Fr). All these six elements are called alkali metals. These elements are placed in the vertical column on the left-hand side of the periodic table. Was this answer helpful? 4.5 (1) (6) (0)
Learn More
Why Do elements in group 1 react with elements in group 7 ... - Answers
Best Answer. Copy. Elements in group 1 have 1 electron in their outer shell. Group 7 electrons have 7 electrons in their outer shell. This means that the group 1 element needs to give its electron
Learn More
What are the Group 1 elements called? - Q & A - Byju's
What are the Group 1 elements called? Francium(Fr). All these six elements are called alkali metals. These elements are placed in the vertical column on the
Learn More
General Chemistry/Chemistries of Various Elements/Group 1
General Chemistry/Chemistries of Various Elements/Group 1 ; Book Cover · Introduction · v • d • e ; Units: Matter · Atomic Structure · Bonding · Reactions ·
Learn More
The Periodic Table of Elements - Elements in Group 1 - Mr Ruel Tuition
Group 1 Elements The elements in Group 18 are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr). Group 1 elements are: also known as alkali metals (see why under chemical properties) shiny when cut, but quickly tarnished upon exposure to air soft and can be cut with a knife
Learn More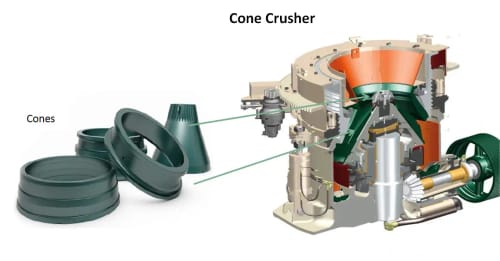
nuspec File Reference for NuGet | Microsoft Learn
The

Spare and Wear Parts for Mining Equipment ᐉ Element Global
Element Group manufactures spare and wear parts under its own brand Element® in accordance with ISO 9001 standard. We develop, design, and manufacture parts for crushing, screening, and slurry handling equipment, in addition to conveyor components, wear-resistant materials, and mill linings. We also supply general-purpose industrial parts
Learn More
Group 1A — The Alkali Metals - The Parts of the Periodic Table
Group 1A (or IA) of the periodic table are the alkali metals: hydrogen (H), lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (
Learn More
Group 1 Elements - Alkali Metals, Properties, Trends, Uses - ProtonsTalk
Group-1 Elements of the Modern Periodic Table consist of Hydrogen (H), Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Caesium (Cs), and Francium (Fr). Out of these elements except for hydrogen, the remaining elements are popularly known as the Alkali metals. Index History Properties of Group 1 Elements or Alkali Metals Physical Properties
Learn More
Group 1 and group 2: Alkali and alkaline earth metals
One of the most common reactions with group 1 and 2 elements is the reaction with water. · Another common reaction is the group 1 or 2 reaction with oxygen. · The
Learn More
THE s-BLOCK ELEMENTS - NCERT
10.1 GROUP 1 ELEMENTS: ALKALI. METALS. The alkali metals show regular trends in their physical and chemical properties with the increasing atomic number.
Learn More
Group 1 Elements Flashcards | Quizlet
Group 1 metals are a lot MORE REACTIVE than transition metals- they react more vigorously with water, oxygen or group 7 elements. Group 1 metals are also a lot LESS DENSE, STRONG and HARD than the transition metals, and have much LOWER MELTING POINTS.
Learn More
Hillsboro, Oregon Platinum Group Elements (PGE) Mines
Filter 1 platinum group elements (pge) mines by commodity, disposition, development status, and record type in Hillsboro, Oregon. Quick Facts Aluminum , Free Water , Iron , Mercury , and Mineral Pigments mines located in Hillsboro, Oregon.
Learn More
PDF Group 1 (1A) - Alkali Metals - umb.eduPDF
element unique chemistry. Because beryllium's chemistry is so different from the other group 2 elements, the term "alkaline earth" is usually restricted to Mg, Ca, Sr, Ba, Ra.!Mg2+ has a similar charge density to Li+, so the two elements show some similar chemistry (e.g., tendency to form useful organometallic compounds).
Learn More
2.5 The Periodic Table – Chemistry - IU Pressbooks
Groups are numbered 1–18 from left to right. The elements in group 1 are known as the alkali metals; those in group 2 are the alkaline earth metals; those in 15
Learn More
Group 1 and Group 2 Elements on the Periodic Table
Group one element shares common characteristics. they're all soft, silver metals. because of their low ionisation energy. Group 2 elements are low density,
Learn More
What is the name of the Group 1 elements in the periodic table?
Main group 1 (1A) of the periodic table is given the name of alkali metals. This is the group that begins with lithium (Li) and sodium (Na).
Learn More
Grouping the Elements - Hilldale.k12.ok.us
Alkali metals are the most reactive metals because their atoms can easily give away the one outer-level electron. Page 3. II. Group 2: Earth. Metals. • Less
Learn More